Language Learning for Pharmaceutical Professionals
Trusted by GSK and leading pharmaceutical companies. Our AI-powered platform helps Ontario teams collaborate fluently with Quebec and global colleagues—from sales meetings to medical presentations.
Trusted by leading organizations
Key results after just a few months
Measurable impact on confidence, collaboration, and daily language use.
Confidence Improvement
Noticed an improvement in their confidence
Cross-site Collaboration
Felt more confident participating in cross-site collaboration
More French at Work
Used more French at work after the pilot
Why growing organizations choose Conversaflex
Hear from leaders who transformed their teams' language skills with Conversaflex.
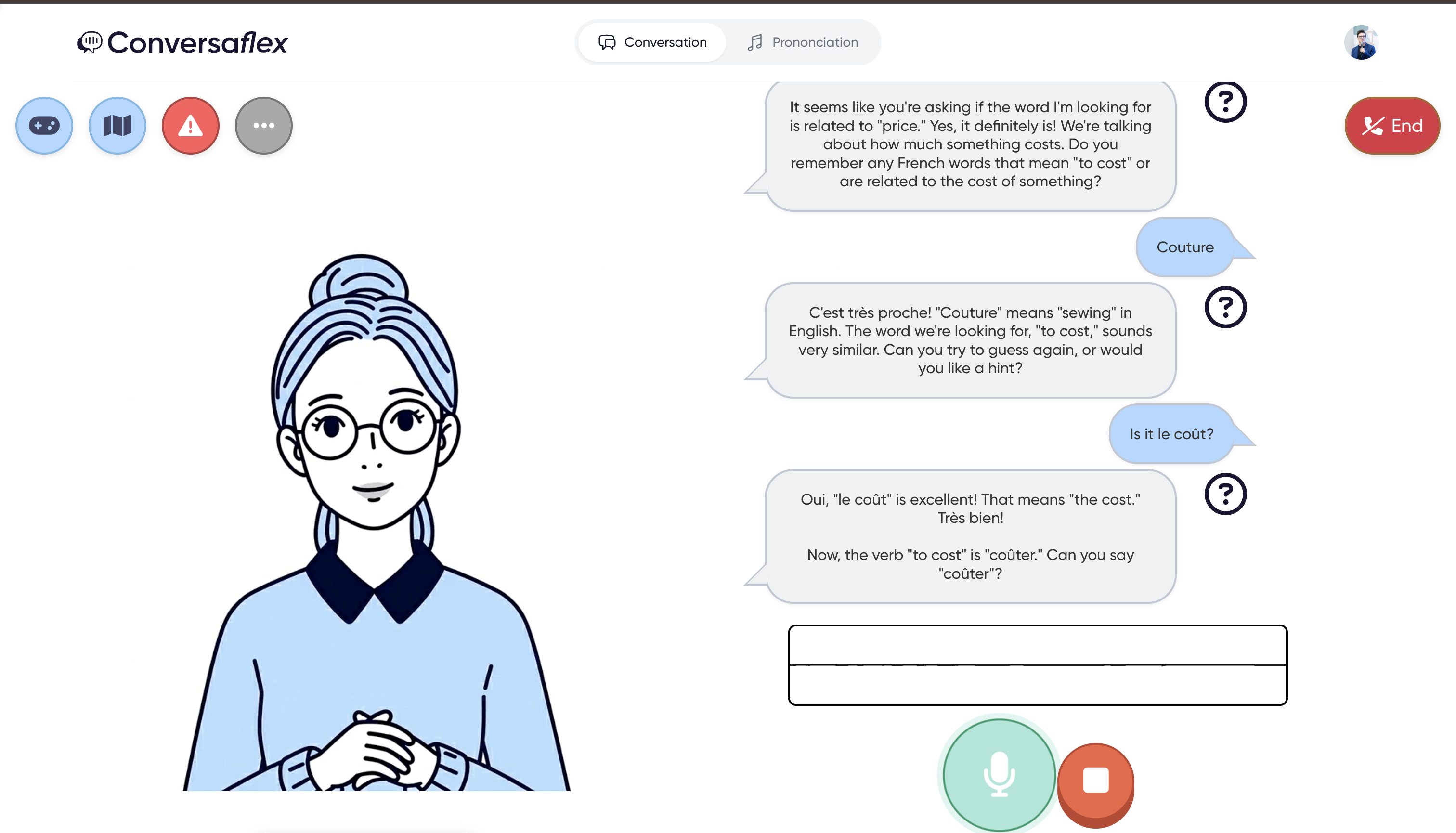
Conversational breakthrough
What Conversaflex gave me is the conversation. That's what I was really missing to make it to the next level of speaking and understanding French.
Rebecca
Asset Laboratory Study Manager
Standing ovation
I was able to share full sentences about our sales performance, which was met with a round of applause from the Quebec team.
Nick
Marketing Director
Medical presentations
Being able to understand what the physicians were saying and understand the presentation was really eye-opening. I wouldn't have been able to do that before the program.
Kathy
Medical Science Liaison
FAQ
How does Conversaflex help pharmaceutical companies comply with Quebec's Bill 96 language requirements?
What French vocabulary do pharmaceutical professionals learn with AI-powered training?
Can pharmaceutical teams practice French for regulatory submissions and documentation?
How quickly can pharma employees achieve workplace French proficiency for Quebec operations?
The learner journey
Every session is a real conversation — spoken, corrected, and adapted in real time.